Specialized Programs
Myocarditis
Learn more
Myocarditis is inflammation of the heart muscle (myocardium). Myocarditis can result from infection (for example, a virus), autoimmunity (when the immune system attacks one’s own body) as well as drugs including cancer immunotherapies and vaccines. UCSF has developed a center of excellence in myocarditis and other inflammatory cardiovascular diseases. We systematically study these patients and collaborate with other academic centers and industry to develop better diagnostic and treatment strategies for patients.
Filamin C (FLNC)
Learn More
Filamin C (FLNC) is a type of filamin protein that is mainly found in skeletal and heart muscle cells, and it helps maintain stability of the muscle. There are some genetic variants (or mutations) that affect the FLNC gene that encodes for filamin C. These mutations can cause abnormal filamin C proteins, which in turn can lead to dysfunction in the heart. The UCSF FLNC Cardiomyopathy program aims to better understand how FLNC variants affect the heart in patients who have these mutations.
Clonal Hematopoiesis of Indeterminate Potential (CHIP)
Learn more
Clonal hematopoiesis of indeterminate potential (CHIP) occurs when somatic mutations occur in blood cells. These mutations do not directly cause cancer per se but they do lead to clonal expansion of cells and occur more commonly as patients age and may result in cancer. It is now known that patients with CHIP are especially at risk for heart disease including acute heart attack and stroke. The UCSF CHIP clinic is developing ways to screen for, diagnose and treat CHIP patients.
Cardiac Amyloidosis Program
Learn More
Cardiac amyloidosis is an increasingly important but under-recognized heart disease that results from the uncontrolled deposition of amyloid fibrils in the heart. Patients with cardiac amyloidosis can develop progressive heart failure, conduction disease, reduced quality of life, and early mortality.
At the UCSF Cardiac Amyloidosis Program, we offer comprehensive, patient centered care for all forms of cardiac amyloidosis. We have developed protocols for diagnosis, treatment, and monitoring of patients who are referred to our clinic.
Carcinoid Heart Disease
Learn More
Neuroendocrine tumors (NETs) are rare, slow growing tumors that can be located anywhere in the body, but most commonly occur in the abdomen. These tumors can secrete hormones which can result in patients having specific symptoms such as flushing, diarrhea, and abdominal cramping (carcinoid syndrome). The hormones can also reach the heart and can lead to heart valvular disease. Occasionally patients have minimal symptoms. But, in many cases, the patient may have severe valve disease that can result in heart failure, necessitating valve replacement surgery.
As a part of the UCSF Center for Neuroendocrine Tumors, we work closely with a multi-disciplinary team including oncologists, interventional radiologists and cardiac surgery offering comprehensive evaluation and treatment plans for carcinoid heart disease.
Pericarditis
Learn more
Pericarditis is inflammation of the pericardium, the heart's outermost layer. Pericarditis can result from various factors such as infections, post-cardiac injury syndrome, autoimmune disease, cancer, and cancer treatments. At UCSF, we have developed a center of excellence in pericarditis, along with other inflammatory cardiovascular diseases. Our approach involves thorough patient evaluation and collaboration with leading academic institutions to advance diagnostic and treatment options.
Hereditary Hemorrhagic Telangiectasia (HHT)
Learn More
Hereditary Hemorrhagic Telangiectasia is a genetic disease where patients develop abnormal and fragile blood vessels between arteries and veins (arteriovenous malformations or AVMs). HHT affects approximately 1 and 5000 individuals, and commonly manifests with nosebleeds. Patients with HHT will may also develop AVMs in in the brain, lungs, and/or liver.
Liver AVMs are seen up to 75% of individuals with HHT. Over time, if left unmonitored and untreated, the high output in the circulation may lead to heart failure. As part of the UCSF HHT Center of Excellence, we partner with interventional radiologists, hematologists, and liver specialists designing novel protocols for the diagnosis and management of heart disease in patients with HHT.

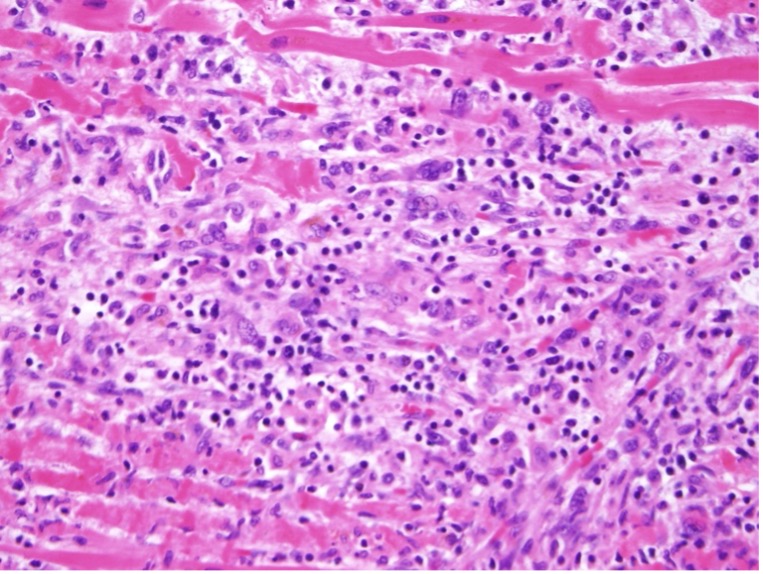 Image:
Image: 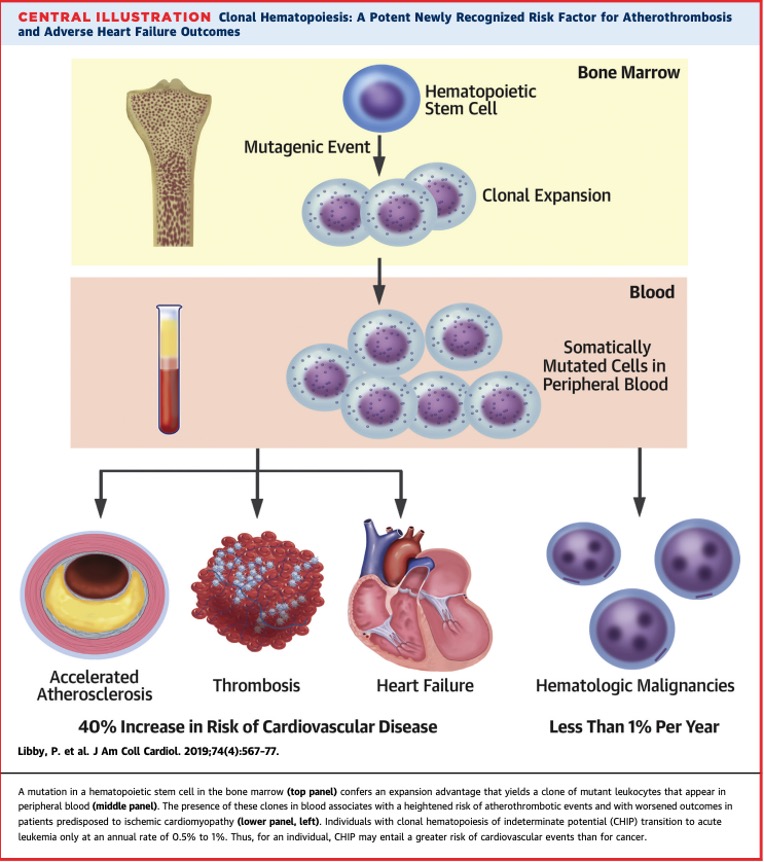 Image:
Image: