Myocarditis Research at UCSF
Research is central to everything we do at the UCSF Myocarditis Center. Our programs span fundamental basic science, translational investigation, and clinical research. These efforts all have a single unified goal: to understand why myocarditis happens, who is most at risk, and how we can detect and treat it earlier and more effectively. We are not simply studying a disease; we are building an entirely new framework for understanding how the immune system and the heart interact, and what happens when that relationship goes wrong.
Understanding the Biology of Myocarditis
At the heart of our research is a fundamental question: what causes the immune system to turn against the heart? In most cases of myocarditis, the immune system (designed to protect us from viral and bacterial infections) becomes the instrument of injury, dispatching specialized cells into cardiac tissue, where they attack and destroy heart muscle cells.
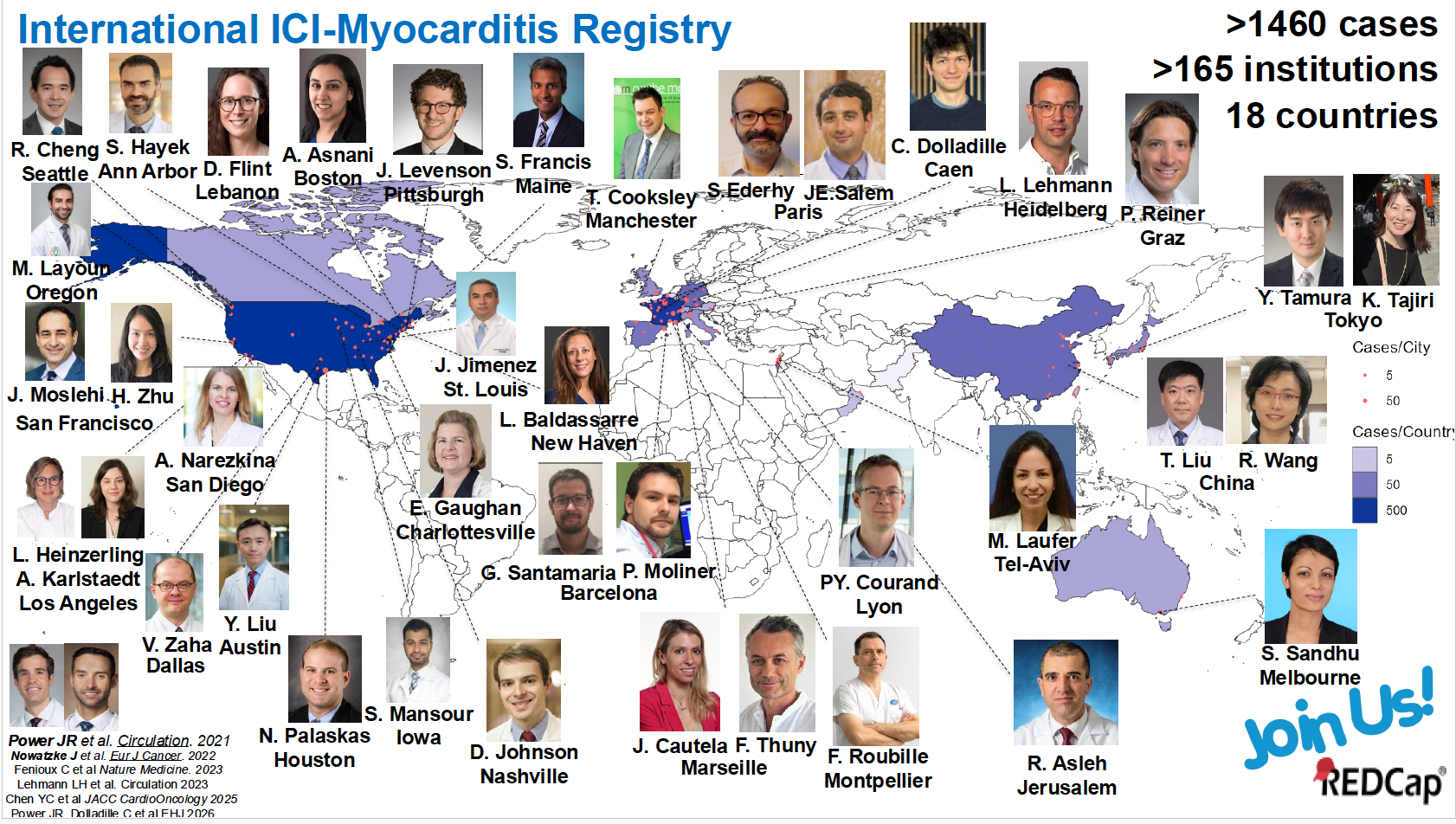
To study this process with precision, the laboratories of Dr. Javid Moslehi and Arun Padmanabhan have developed a series of mouse models that faithfully replicate immune injury patterns seen in human myocarditis. One particularly dangerous form is triggered by an immune checkpoint inhibitor (ICI). ICIs are among the most transformative advances in cancer treatment in the past decade, harnessing the immune system to fight tumors. However, in roughly one percent of patients, they trigger a severe and often fatal form of myocarditis. Our cardio-oncology group initially discovered this syndrome in 2016. To better understand how ICI-myocarditis occurs, the UCSF team has led an international effort to characterize the syndrome, creating a registry of cases from around the world spanning more than 15 countries and nearly 200 centers. With mortality rates approaching forty percent in some studies, ICI-myocarditis is the most lethal immune-related complication of cancer therapy. Our group has been at the forefront of defining this syndrome, both clinically and mechanistically, and is widely recognized as one of the world’s leading research programs on this topic.
Separately, the mouse (and other pre-clinical) models that our research program has created allow us to observe the disease process unfold in real time at the cellular and molecular levels. Using single-cell RNA sequencing, a technology that reads the activity of thousands of individual genes in thousands of individual cells simultaneously, we have mapped exactly which immune cell populations expand during myocarditis and what those cells are doing. At the same time, we have been able to show which potential target genes/proteins the activated immune cells attack or why males and females are affected differently by myocarditis.
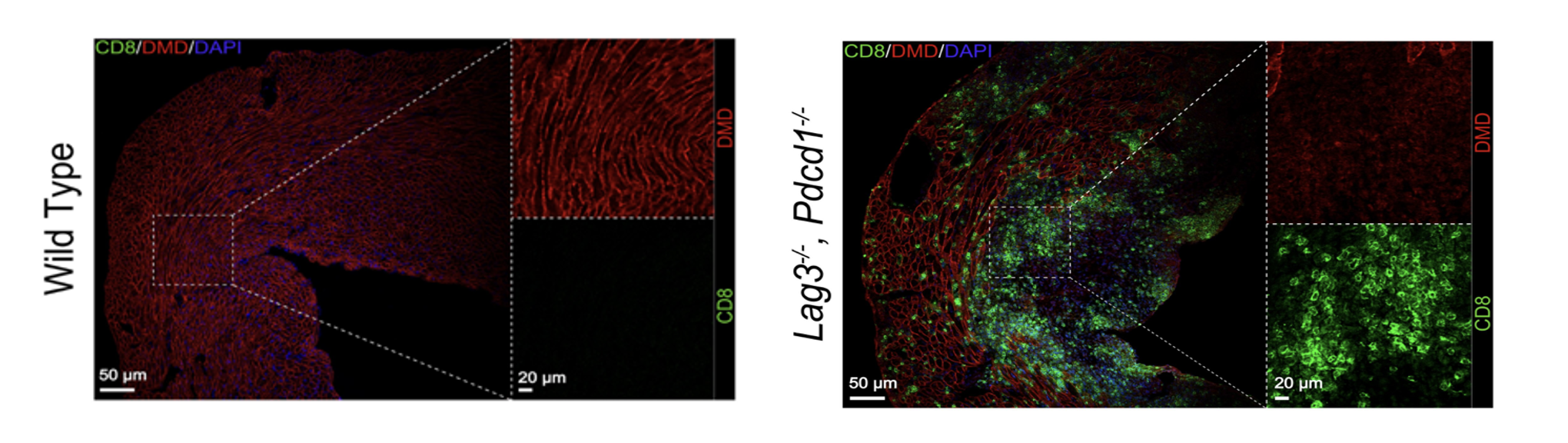
What makes this research program exceptional is the complementary depth that Drs. Moslehi and Padmanabhan each bring to it. Dr. Moslehi is a physician-scientist who has spent his career at the intersection of immunology and cardiovascular medicine, developing and characterizing mouse models that form the foundation of this work and building clinical programs that connect laboratory discoveries to patient care. Dr. Padmanabhan brings world-class expertise in single-cell biology and the molecular crosstalk between different cell types, the complex signaling conversations that happen between immune cells, heart muscle cells, and the fibroblasts that drive scarring. Together, their laboratories are uniquely equipped to ask not just which cells are present in the inflamed heart, but what those cells are saying to each other, how the heart is responding, and where in that cascade of signals a therapeutic intervention might have the greatest impact. It is a collaboration that combines the right disease models, the right molecular tools, and the right clinical context, generating insights into myocarditis biology that neither laboratory could produce alone.
Discovering the Next Generation of Biomarkers
One of the most pressing problems in myocarditis is that we are flying blind diagnostically. Troponin, the standard blood test for heart injury, rises whenever heart muscle cells die, whether from a heart attack, myocarditis, or any number of other causes. It tells us something is wrong, but not what or why. Echocardiography often appears normal even in seriously ill patients. Cardiac MRI, the most sensitive conventional imaging tool, misses the diagnosis in more than half of biopsy-confirmed cases. This diagnostic gap costs lives. We are tackling this problem with a technology called Bio-Orthogonal Non-Canonical Amino Acid Tagging (BONCAT). BONCAT represents a fundamentally new approach to biomarker discovery, and its application to myocarditis is one of the most innovative aspects of our research program.
are flying blind diagnostically. Troponin, the standard blood test for heart injury, rises whenever heart muscle cells die, whether from a heart attack, myocarditis, or any number of other causes. It tells us something is wrong, but not what or why. Echocardiography often appears normal even in seriously ill patients. Cardiac MRI, the most sensitive conventional imaging tool, misses the diagnosis in more than half of biopsy-confirmed cases. This diagnostic gap costs lives. We are tackling this problem with a technology called Bio-Orthogonal Non-Canonical Amino Acid Tagging (BONCAT). BONCAT represents a fundamentally new approach to biomarker discovery, and its application to myocarditis is one of the most innovative aspects of our research program.
Here is the core idea: every protein in the body is built from amino acids, the molecular building blocks of life. BONCAT exploits this by introducing a synthetic amino acid, one that does not naturally exist in biology, into living cells. Cells that have been genetically engineered to accept this synthetic amino acid incorporate it into every new protein they build, tagging those proteins with a chemical handle that can later be grabbed, isolated, and identified. 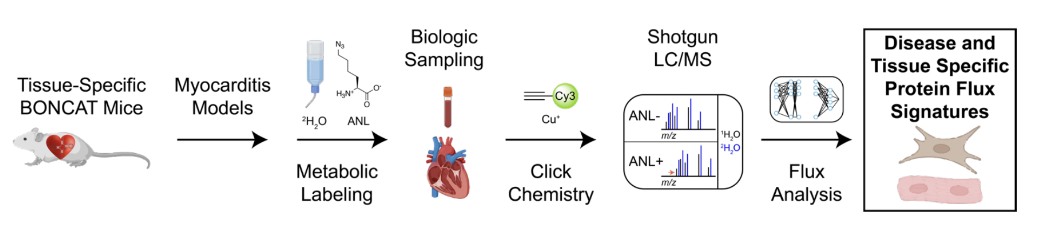
The power of this approach lies in its specificity. By restricting which cells can incorporate the synthetic amino acid, targeting it exclusively to heart muscle cells, for instance, or exclusively to immune cells, Drs. Moslehi and Padmanabhan's team can identify proteins that are being actively produced by those specific cell types during injury and released into the bloodstream. This has never been done before in the context of myocarditis. Traditional biomarker discovery involves taking a blood sample from a sick patient and trying to find something unusual in the sea of thousands of circulating proteins, with no way to know where any of them came from. BONCAT solves what scientists call the tissue-of-origin problem. Every candidate biomarker identified comes with a known address: we know exactly which cell made it, and when.
Proteins identified in the mouse models as promising candidates are screened against blood samples from our clinical biorepository. Candidates that distinguish the two groups, and that differ from the patterns seen in ordinary heart attacks, advance toward clinical validation. The goal is a blood test that can tell a physician not just that the heart is under attack, but also what kind of attack it is and where it is coming from, enabling faster, more targeted treatment.
Imaging Inflammation and Fibrosis in the Heart
Even with better biomarkers, there is something a blood test cannot do: show you where in the heart the injury is happening, how extensive it is, and what biological process is driving it. For that, we need imaging. And the imaging tools we currently have are, frankly, inadequate for myocarditis.
Working in close collaboration with colleagues in radiochemistry and nuclear medicine, Drs. Moslehi and Padmanabhan are developing and testing a new generation of PET imaging tracers to visualize the biological processes driving heart injury. The technology behind these tracers is called Restricted Interaction Peptides, or RIPs. RIPs are engineered molecules with a clever built-in mechanism: they are biologically inert until they encounter a specific target protein, at which point they are activated and lock onto nearby cell membranes, generating a concentrated imaging signal precisely where that target protein is active. This design provides extraordinary specificity: the tracer lights up only where the biology of interest is occurring. This platform is being applied to two targets that go to the heart of myocarditis biology. Building on this approach, we have launched a Phase 1 clinical trial to evaluate these novel tracers in patients with myocarditis.
Treatment Options
Current treatment options for myocarditis remain uncertain and lack specificity. Management strategies vary based on severity and clinical presentation. While devices may be used to temporarily stabilize very sick patients, there are no approved drugs to treat the underlying immune activation. In fact, it is safe to say that in 2026, there have been no drugs that have bene shown to be effective in treating any form of myocarditis. Physicians might prescribe immunosuppressants such as colchicine or corticosteroids, but studies have failed to show any efficacy.
The UCSF Myocarditis Center is at the forefront of developing targeted therapies for myocarditis. One promising treatment is Ruxolitinib, a nonselective Janus kinase (JAK) inhibitor that dampens inflammatory pathways that drive heart damage. Our team recently published a Circulation study highlighting the successful use of Ruxolitinib in a patient with acute fulminant myocarditis, resulting in rapid improvement in cardiac function and recovery from cardiogenic shock. Single-cell profiling provided insights into how inhibiting JAK-mediated inflammatory pathways may reverse immune-driven injury to the heart. These developments offer new hope for patients with severe myocarditis and underscore the importance of ongoing research into targeted, personalized therapies. In addition, in cases of ICI-myocarditis, the combination of Ruxolitinib and another T-cell inhibitor – abatacept - has shown efficacy.
Clinical Research Programs and Biorepositories
The basic science work of Drs. Moslehi and Padmanabhan are anchored by a robust clinical research infrastructure that gives laboratory discoveries an immediate pathway to human validation.
INFLAME Biorepository
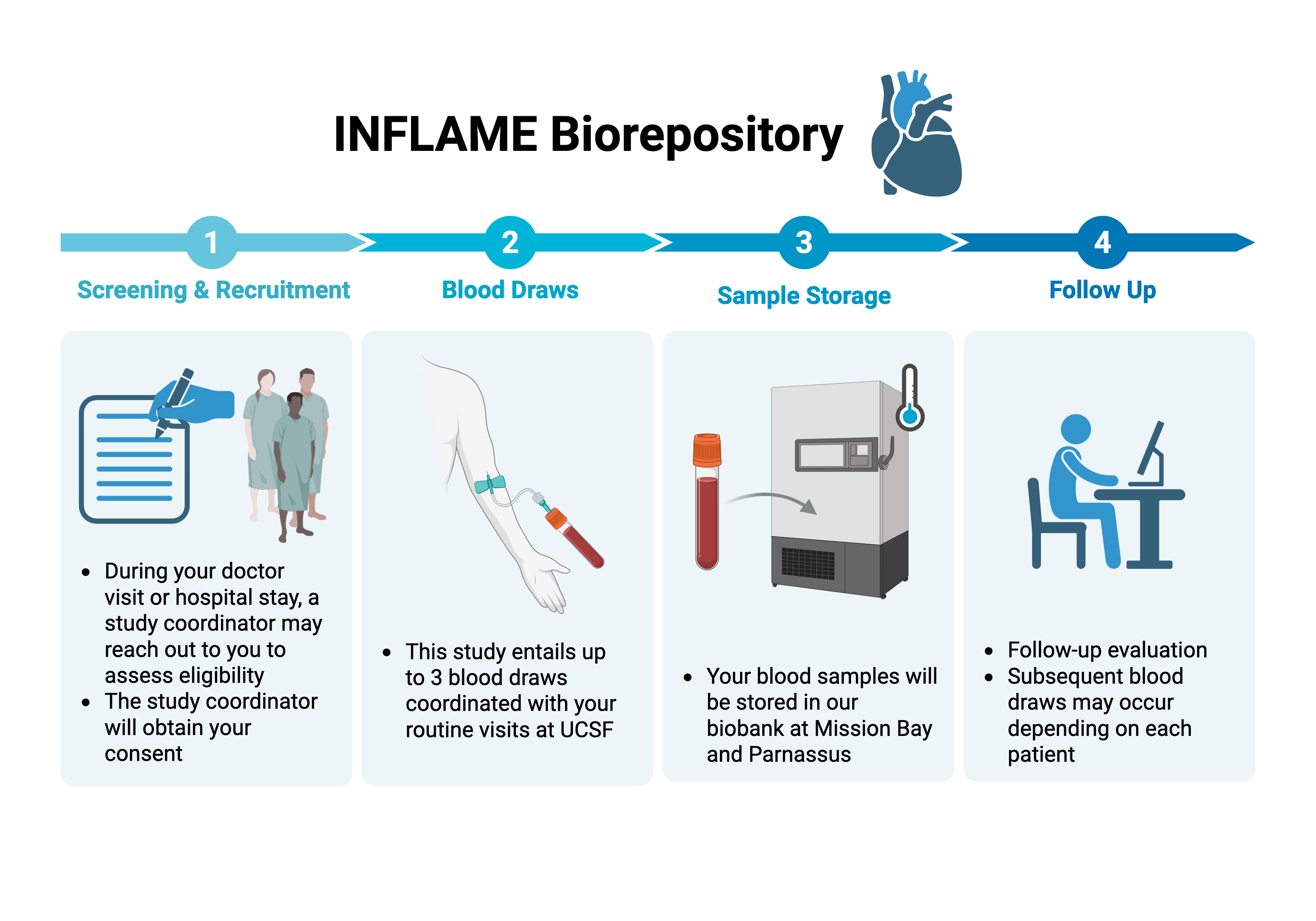 A prospective biobank systematically collecting blood, tissue, and clinical data from patients with myocarditis and related inflammatory heart conditions, including pericarditis and endocarditis. This resource links biological samples to detailed clinical outcomes, allowing us to test whether what we discover in the laboratory actually predicts patient outcomes.
A prospective biobank systematically collecting blood, tissue, and clinical data from patients with myocarditis and related inflammatory heart conditions, including pericarditis and endocarditis. This resource links biological samples to detailed clinical outcomes, allowing us to test whether what we discover in the laboratory actually predicts patient outcomes.
Cardio-Oncology and Immunology Biorepository
Focused specifically on the intersection of cancer treatment and cardiovascular disease, this biorepository supports research into how immunotherapies and targeted cancer drugs affect the heart, and who is most vulnerable to those effects.
International ICI-Myocarditis Registry
UCSF is a leading contributor to this global registry, which aggregates data from patients around the world who have developed myocarditis as a complication of immune checkpoint inhibitor therapy. By pooling data across institutions and countries, the registry has the statistical power to answer questions about susceptibility, risk factors, and outcomes that no single center could address on its own.
Together, these resources mean that when the Moslehi or Padmanabhan laboratories identify a promising biomarker or imaging signal in a mouse model, we have the patient cohorts, the clinical data, and the institutional infrastructure to test it in humans rapidly and rigorously, thereby closing the loop between discovery and impact.
